|
3/10/2024 0 Comments Periodic table reactivity trends Some relevant data to the reactivity might be the ionisation energies of the elements. Chemists try to use real data where possible to inform their ideas. For example, a quick glance at Figure AT5.2. Student worksheet 18SW Trends in reactivity in the Periodic Table of 3 Chemistry for the gifted and talented. These are homework exercises to accompany the Textmap created for 'Chemistry: The Central Science' by Brown et al. by Brown, LeMay, Bursten, Murphy, and Woodward. That way, we can get a better look at the relationship. A general chemistry Libretexts Textmap organized around the textbook. Often it is useful to plot data on a graph. As a result, fluorine is usually thought of as the most electronegative element. However, on many scales, fluorine would be the most electronegative atom here. A collection of visually stimulating and informative infographics about the elements, which would make a valuable addition to any science classroom. These can be found in nature in both free and combined states. 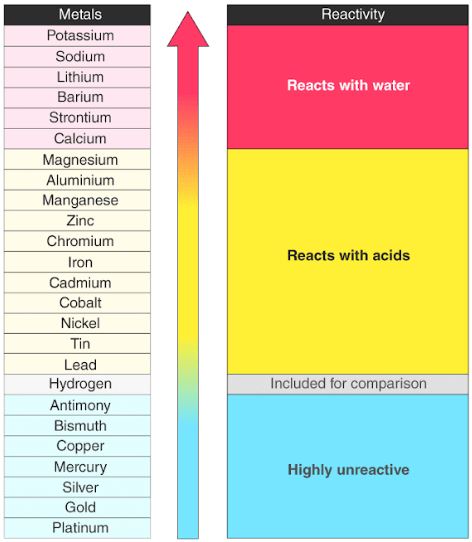
It provides a systematic framework for comparing and. 
The reactivity series is a hierarchical arrangement of elements based on their relative tendency to undergo chemical reactions. It consists of the elements oxygen, sulfur, selenium, tellurium and polonium. Interactive Periodic Table Periodic Trends Made Easy Atomic Radius Trend Ionization Energy Trend Electronegativity Trends Defining the Reactivity Series. As a result, noble gases are also given electronegativity values on this scale. The oxygen family, also called the chalcogens, consists of the elements found in Group 16 of the periodic table and is considered among the main group elements. The Allen scale just depends on the ability of an atom to interact with light, which is something even noble gases can do. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. Some electronegativity scales do not have values for the noble gases, because they are based on experimental measurements of compounds, and noble gases do not commonly form compounds with other elements. The trends in the periodic table are generally the radius of the atom gets bigger or the radius of the atom gets smaller sort of thing. \): The Allen electronegativity values of the second-row elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
